
Using very powerful electromagnets, he observed that the canal rays deflected in a direction opposite to the cathode rays, which implied they carried a positive charge of electricity, and this led them to be renamed as 'positive rays'. One year later, in 1898, Wilhem (Willi) Wien, while working as an assistant to Hermann Helmholtz in Berlin, was inspired by Thomson's work and began his own investigations into cathode and canal rays. Source: © National Library of Congress/SPL Using the strongest magnet he had, one that certainly had an effect on the cathode rays, Goldstein attempted to deflect his canal rays, but he observed no change in path. 2a Unlike the cathode rays, the colour of the canal rays depended on the identity of the residual gas in the tube, though the significance of this observation was not appreciated at the time. On account of their appearance, Goldstein named them Kanalstrahlen, which means 'canal rays'. 
If the cathode was relatively thick, they would appear as a parallel beam. Some years later, in 1886, the German physicist Eugen Goldstein noticed that if he used a perforated cathode, then in addition to the cathode rays between the anode and the cathode, there was a different type of ray emerging from the holes in the cathode, and moving in the opposite direction to the cathode rays. 1 This was early evidence of 'cathode rays', though nothing was known of their properties. In 1858, professor of physics at the University of Bonn, Julius Plücker, while investigating the action of a magnet on the electric discharge of rarefied gases, observed a cathode-induced green fluorescence on the glass walls of a discharge tube. The phenomenon began to receive rigorous scientific study in the mid-19th century with the advent of induction coils that could provide high voltages sustained over long periods of time and the availability of high capacity batteries to power the induction coils. To connect the tube in different way's to a galvanometer the positive or negative charged particles can be measured by experiment.ġ8 Canal rays can be seen in the left of the picture.At the end of the 18th century experimental scientists were aware that a spark generated electrostatically could travel longer distances in a partially evacuated glass tube than in air. This tube has some extra electrodes compared to the Goldstein Canal Ray tube. Wien did several experiments from, his experiments were similar to JJ.Thomson almost the same time. Goldstein (1886) found that some rays passed through these holes in a direction opposite to that of the cathode rays.ġ6 Wien's Canal Ray tube Wien's Canal Ray tube is named after it's inventor Wilhelm Wien. By using a discharge tube containing a perforated cathode. Hence after the discovery of the negatively charged constituent (electron) of an atom, attempts were made to discover the positively charged counterpart of electrons. 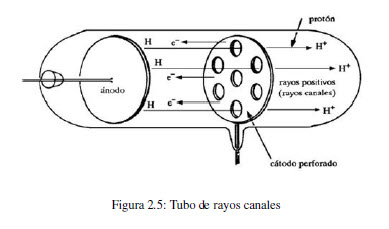
Because these rays pass through the holes, or channels, in the cathode, Goldstein called them canal rays.ĩ DISCOVERY OF PROTON POSITIVE RAYS OR CANAL RAYSĪtoms are electrically neutral. Goldstein concluded that in addition to the electrons, or cathode rays, that travel from the negatively charged cathode toward the positively charged anode, there is another ray that travels in the opposite direction, from the anode toward the cathode. Since these canal rays travel in the opposite direction from the cathode rays, they must carry the opposite charge.Ĩ Canal Rays In 1886 Eugen Goldstein noted that cathode-ray tubes with a perforated cathode emit a glow from the end of the tube near the cathode. Goldstein could not explainthis phenomenon, it took 12 years before Goldstein's paper was published.Ĭanal Rays or positive Protons (red glow) Electrons in the lower partħ Canal Rays When the cathode of a cathode-ray tube was perforated, Goldstein observed rays he called "canal rays," which passed through the holes, or channels, in the cathode to strike the glass walls of the tube at the end near the cathode. The electrons in the lower part of the tube can be deflected bya magnetic field but the canal rays almost not. In fact these are positively charged protons, producing a reddish light in the upper part of the tube while in the lower part the usual green emission of electrons can be seen when they hit the glass wall. Discovered in 1886 by Eugen Goldstein ( ) who named this"canal rays". This tube demonstrates that besides the cathode rays there is another stream that travels in the opposite direction as the electron flow. 
Presentation on theme: "The Goldstein Canal Ray Tube This tube demonstrates that besides the cathode rays there is another stream that travels in the opposite direction as."- Presentation transcript:
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
